A) Pb.
B) Ag.
C) Sn.
D) Mg.
E) Cu.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the following redox equation. Mn(OH) 2(s) + MnO4-(aq) → MnO42-(aq) (basic solution) When the equation is balanced with the smallest whole number coefficients,what is the coefficient for OH-(aq) and on which side of the equation is OH-(aq) present?
A) 4,reactant side
B) 4,product side
C) 6,reactant side
D) 6,product side
E) 8,reactant side
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The voltaic cell composed of Co(s) ,Co2+(aq) ,Cu(s) ,and Cu2+(aq) has E°cell = 0.62 V.If E° of the cathode half-cell is 0.34 V,what is E° of the anode half-cell? Cu2+(aq) + Co(s) → Cu(s) + Co2+(aq)
A) -0.28 V
B) -0.96 V
C) 0.28 V
D) 0.96 V
E) 0.21 V
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What would you observe if you set up the following electrochemical cell: Ag|AgNO3(0.001 M) ||AgNO3(1 M) |Ag?
A) Electrons will flow from left to right,causing a decrease in the AgNO3 concentration in the left cell,and an increase in the AgNO3 concentration in the right cell.
B) Electrons will flow from right to left,causing an increase in the AgNO3 concentration in the left cell,and a decrease in the AgNO3 concentration in the right cell.
C) Electrons will flow from left to right,causing an increase in the AgNO3 concentration in the left cell,and a decrease in AgNO3 concentration in the right cell.
D) Electrons will flow from right to left,causing a decrease in the AgNO3 concentration in the left cell,and an increase in the AgNO3 concentration in the right cell.
E) There will be no electron flow because the reduction potential at both electrodes is the same.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
True/False
In the electrolyte of an electrochemical cell,current is carried by electrons moving from the anode to the cathode.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A cell can be prepared from copper and tin.What is the E°cell for the galvanic cell that forms from the following half-reactions? Cu2+(aq) + 2e-  Cu(s) E° = 0.34V
Sn4+(aq) + 2e-
Cu(s) E° = 0.34V
Sn4+(aq) + 2e-  Sn2+(aq) E° = 0.13V
Sn2+(aq) E° = 0.13V
A) 0.47 V
B) 0.21 V
C) -0.21 V
D) -0.47 V
E) 0.42 V
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is E°cell for the following reaction,and is it spontaneous or nonspontaneous under standard-state conditions? 2Fe3+(aq) + 2H2O(l) → H2O2(aq) + 2H+(aq) + 2Fe2+(aq)
Given:
H2O2(aq) + 2H+(aq) + 2e-
 2H2O(l) E° = 1.77V
Fe3+(aq) + e-
2H2O(l) E° = 1.77V
Fe3+(aq) + e-  Fe2+(aq) E° = 0.77V
Fe2+(aq) E° = 0.77V
A) E°cell = -1.00 V,nonspontaneous
B) E°cell = -1.00 V,spontaneous
C) E°cell = 1.00 V,nonspontaneous
D) E°cell = 1.00 V,spontaneous
E) E°cell = -0.23 V,nonspontaneous
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the oxidizing agent in the (unbalanced) reaction? Cu(s) + H+(aq) + NO3-(aq) → NO(g) + H2O(l) + Cu2+(aq)
A) Cu
B) H+
C) NO3-
D) NO
E) Cu2+
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When the following redox equation is balanced with the smallest whole number coefficients,the coefficient for the hydrogen sulfate ion will be ______. Al(s) + HSO4-(aq) + OH-(aq) → Al2O3(s) + S2-(aq) + H2O(l)
A) 1
B) 3
C) 4
D) 6
E) 8
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the equilibrium constant at 25°C for the following reaction? (R = 8.314 J/K • mol,F = 96,500 C • mol-1)
Sn2+(aq) + Fe(s)  Sn(s) + Fe2+(aq) E°cell = 0.30V
Sn(s) + Fe2+(aq) E°cell = 0.30V
A) 1.2 × 105
B) 1.4 × 1010
C) 8.6 × 10-6
D) 7.1 × 10-11
E) 2.3 × 1023
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A voltaic cell consists of a Hg/Hg22+ electrode (E°= 0.85 V) and a Sn/Sn2+ electrode (E°= -0.14 V) .Calculate [Sn2+] if [Hg22+] = 0.24 M and Ecell = 1.04 V at 25°C.
A) 0.00010 M
B) 0.00070 M
C) 0.0049 M
D) 0.030 M
E) 0.050 M
G) C) and D)
Correct Answer

verified
Correct Answer
verified
True/False
At equilibrium E° = 0.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Complete and balance the following redox equation.When properly balanced using the smallest whole-number coefficients,the coefficient of S is H2S + HNO3 → S + NO + H2O (acidic solution)
A) 1.
B) 2.
C) 3.
D) 5.
E) 6.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Complete and balance the following redox equation using the set of smallest whole numbers coefficients.What is the sum of the coefficients? HI + HNO3 → I2 + NO (acidic solution)
A) 5
B) 7
C) 14
D) 17
E) None of these choices is correct.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
True/False
The Faraday constant represents the charge of 1 mole of electrons.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
A salt bridge allows movement of cations and anions from one half-cell to the other.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Based on the data presented below,which is the strongest reducing agent? E°(V) Al3+ + 3e- → Al(s) -1.66 AgBr(s) + e- → Ag(s) +Br- +0.07 Sn4+ + 2e- → Sn2+ +0.14 Fe3+ + e- → Fe2+ +0.77
A) Fe3+
B) Fe2+
C) Br-
D) Al3+
E) Al(s)
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Based on the following electrochemical cell,what is the standard reduction potential of metal M at 298 K? 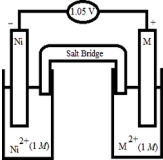 Half-Reaction E° (V) Ni2+(aq) + 2e- →Ni(s) -0.25
Half-Reaction E° (V) Ni2+(aq) + 2e- →Ni(s) -0.25
A) -1.40 V
B) -0.80 V
C) +0.25 V
D) +0.80 V
E) +1.40 V
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is E°cell for a galvanic cell represented by the combination of the following half-reactions? ClO4-(aq) + 8 H+(aq) +8e-
 Cl-(aq) +4H2O(l) E° = 1.389V
VO2+(aq) + 2H+(aq) +e-
Cl-(aq) +4H2O(l) E° = 1.389V
VO2+(aq) + 2H+(aq) +e-
 VO+(aq) +H2O(l) E° = 0.991V
VO+(aq) +H2O(l) E° = 0.991V
A) -0.398 V
B) -2.380 V
C) 0.398 V
D) 2.380 V
E) 6.539 V
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the minimum voltage required for the electrolysis of 1.0 M NaCl in neutral solution? 2H2O + 2Cl- (1.0 M) → H2(1 atm) + Cl2(1 atm) + 2OH-(1 × 10-7 M) Note that 2H2O(l) + 2e- → H2(g) +2OH-(aq) ,E° = -0.83 V, 2H2O(l) + 2e- → H2(g) +2OH-(aq) ,E = -0.41 V([OH-] = 1 × 10-7M) , O2(g) + 4H+(aq) + 4e- →2H2O(l) ,E° = +1.23 V, Cl2(g) + 2e- → 2Cl-(aq) E° = +1.36 V,and Na+(aq) + e- → Na(s) E° = -2.71 V
A) 2.19 V
B) 1.77 V
C) 0.41 V
D) -0.41 V
E) -1.78 V
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 122
Related Exams