A) Because there are more atoms in acetone
B) Because there are more resonance structures for the enolate of acetone
C) It isn't; the allyl anion is less basic.
D) One of the resonance structures for the enolate places the negative charge on the more electronegative oxygen.
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the appropriate sequence of reactions to accomplish the following synthesis. ![Select the appropriate sequence of reactions to accomplish the following synthesis. A) [1] Br<sub>2</sub>, CH<sub>3</sub>CO<sub>2</sub>H; [2] Li<sub>2</sub>CO<sub>3</sub>, LiBr, DMF; [3] CH<sub>3</sub>CH<sub>2</sub>CH<sub>2</sub>CH<sub>2</sub>Br B) [1] Br<sub>2</sub>, CH<sub>3</sub>CO<sub>2</sub>H; [2] Mg, Et<sub>2</sub>O; [3] CH<sub>3</sub>CH<sub>2</sub>CH<sub>2</sub>CH<sub>2</sub>Br C) [1] LDA; [2] BrCH<sub>2</sub>CH<sub>2</sub>CH<sub>2</sub>CH<sub>2</sub>Br; [3] NaOEt D) [1] NaOEt; [2] BrCH<sub>2</sub>CH<sub>2</sub>CH<sub>2</sub>CH<sub>2</sub>Br; [3] LDA](https://d2lvgg3v3hfg70.cloudfront.net/TB5871/11ea9088_707b_6bd2_aec7_b38aab5412d6_TB5871_00_TB5871_00_TB5871_00_TB5871_00_TB5871_00_TB5871_00_TB5871_00.jpg)
A) [1] Br2, CH3CO2H; [2] Li2CO3, LiBr, DMF; [3] CH3CH2CH2CH2Br
B) [1] Br2, CH3CO2H; [2] Mg, Et2O; [3] CH3CH2CH2CH2Br
C) [1] LDA; [2] BrCH2CH2CH2CH2Br; [3] NaOEt
D) [1] NaOEt; [2] BrCH2CH2CH2CH2Br; [3] LDA
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
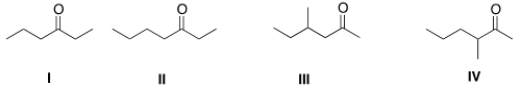
Which of the following four compounds is the most acidic? 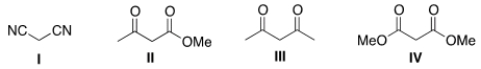
A) I
B) II
C) III
D) IV
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following ketones will give a positive iodoform test?
A) 3-Heptanone
B) 2-Pentanone
C) 3-Hexanone
D) Cyclohexanone
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why can't you prepare 2-tert-butylcyclohexanone by the following reaction? 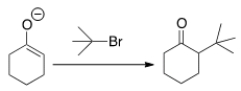
A) Because tert-butyl bromide is too basic.
B) Because tert-butyl bromide cannot undergo an SN2 reaction.
C) Because tert-butyl bromide is a nucleophile.
D) Because tert-butyl bromide is not a stable compound.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is the thermodynamic enolate of 2-methylcyclohexanone? 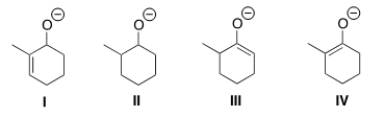
A) I
B) II
C) III
D) IV
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If you want to form a thermodynamic enolate, you want to:
A) Keep the reaction as cold as possible.
B) Use an aprotic solvent such as THF.
C) Use a protic solvent such as ethanol.
D) Use a carboxylic acid.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why is it difficult to stop the halogenation of ketones under basic conditions at the mono-halogenated stage?
A) The ketone undergoes a Bayer-Villigar oxidation.
B) The ketone is reduced.
C) The ketone undergoes an Aldol reaction.
D) The bromine helps to stabilize the second enolate, making the product more acidic than the starting material.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the starting material required to accomplish the following transformation? 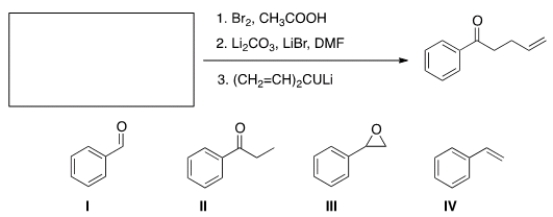
A) I
B) II
C) III
D) IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the major product of the following reaction? 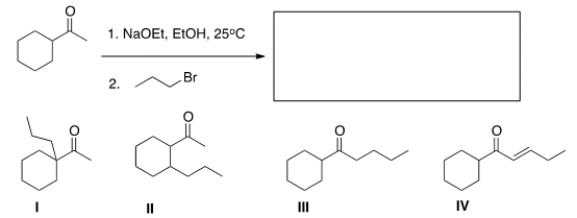
A) I
B) II
C) III
D) IV
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A simple chemical test to distinguish between acetone and 3-pentanone would be the reaction of the compounds with
A) bromine with acetic acid.
B) bromine and aqueous hydroxide ion.
C) THF, LDA at -78 °C followed by reaction with bromine.
D) base and methyl bromide.
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the product of the following reaction? 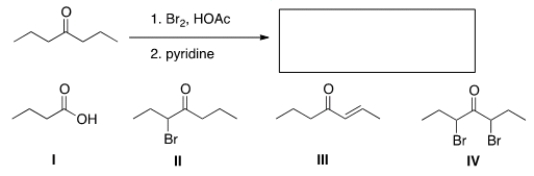
A) I
B) II
C) III
D) IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the missing reagent for the following reaction? 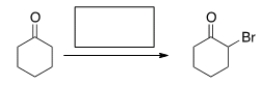
A) Br2/HOAc
B) Br2/KOH
C) Cl2/FeCl3
D) Br2/FeBr3
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is the more stable form of acetophenone? 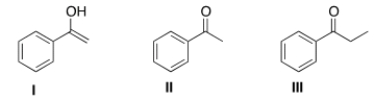
A) Only I
B) Only II
C) Only III
D) I and II are equally stable
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following are enol forms of ethyl acetoacetate drawn below? 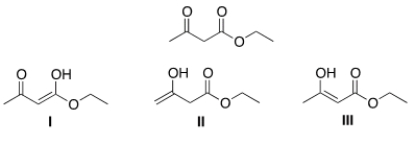
A) Only I
B) Only II
C) Only III
D) I, II, and III
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is (are) the product(s) of the following reaction? 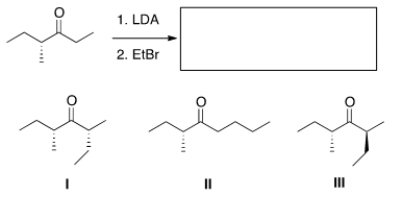
A) Only I
B) Only II
C) Only III
D) Only I and III
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For most compounds with a single keto group in the molecule, equilibrium favors the keto form over the enol form of the compound. This is due largely to what?
A) The C=O bond is much stronger than the C=C bond.
B) The C=C bond is much stronger than the C=O bond.
C) The keto form can undergo intramolecular hydrogen bonding.
D) The enol form can undergo intramolecular hydrogen bonding.
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the product of the following reaction? 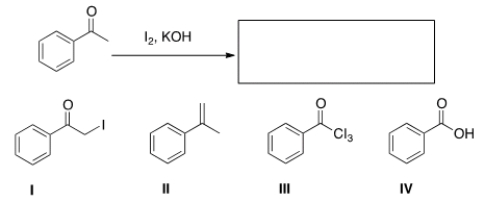
A) I
B) II
C) III
D) IV
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Treatment of 2-hexanone with NaOCH2CH3 followed by CH3Br affords compound X (C7H14O) as the major product. X shows a strong absorption in the IR spectrum at 1713 cm-1, and its 1H NMR data is given below. What is the structure of X? 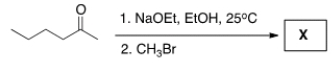
A) I
B) II
C) III
D) IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is the most acidic proton in the following compound? 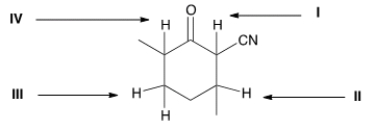
A) I
B) II
C) III
D) IV
F) All of the above
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 41
Related Exams