A) Mn2+(aq) is the oxidizing agent and is reduced.
B) Mn2+(aq) is the oxidizing agent and is oxidized.
C) Mn2+(aq) is the reducing agent and is oxidized.
D) Mn2+(aq) is the reducing agent and is reduced.
E) Manganese does not change its oxidation number in this reaction.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
True/False
If the electrodes of a voltaic cell are connected with an external wire, electrons will flow in this wire from the cathode to the anode.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
In the shorthand notation for cells, a single vertical line represents a salt bridge.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The value of E°cell for the reaction 2Cr3+(aq) + 6Hg(l) 2Cr(s) + 3Hg22+(aq) Is 1.59 V. Calculate G° for the reaction.
A) -921 kJ
B) -767 kJ
C) -460 kJ
D) -307 kJ
E) none of the above
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the reaction CuO(s) + H2(g) Cu(s) + H2O(l) In this reaction, which substances are the oxidant and reductant, respectively?
A) CuO and H2
B) H2 and CuO
C) CuO and Cu
D) H2O and H2
E) none of the above
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the following balanced redox reaction 3CuO(s) + 2NH3(aq) N2(g) + 3H2O(l) + 3Cu(s) Which of the following statements is true?
A) CuO(s) is the oxidizing agent and copper is reduced.
B) CuO(s) is the oxidizing agent and copper is oxidized.
C) CuO(s) is the reducing agent and copper is oxidized.
D) CuO(s) is the reducing agent and copper is reduced.
E) CuO(s) is the oxidizing agent and N2(g) is the reducing agent.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The redox reaction of peroxydisulfate with iodide has been used for many years as part of the iodine clock reaction which introduces students to kinetics. If E°cell = 1.587 V and E° of the cathode half-cell is 0.536 V, what is E° of the anode half-cell? S2O82-(aq) + 2H+ + 2I-(aq) 2HSO4-(aq) + I2(aq)
A) -1.051 V
B) -2.123 V
C) 1.051 V
D) 2.123 V
E) none of the above
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A voltaic cell consists of a Cd/Cd2+ electrode (E° = -0.40 V) and a Fe/Fe2+ electrode (E° = -0.44 V) . If Ecell = 0 and the temperature is 25°C, what is the ratio [Fe2+]/[Cd2+]?
A) 2 × 101
B) 1 × 101
C) 1
D) 1 × 10-1
E) 5 × 10-2
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Examine the following half-reactions and select the strongest oxidizing agent among the species listed.
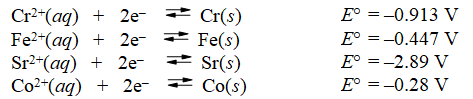
A) Cr2+(aq)
B) Fe(s)
C) Fe2+(aq)
D) Sr2+(aq)
E) Co2+(aq)
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
In the shorthand notation for cells, a double vertical line is used to separate the reduced and oxidized forms of a redox couple.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the E°cell for the cell represented by the combination of the following half-reactions? 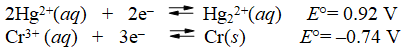
A) -0.18 V
B) 0.18 V
C) 1.28 V
D) 1.66 V
E) 2.12 V
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Examine the following half-reactions and select the weakest oxidizing agent among the species listed.
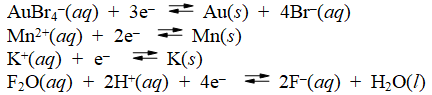
A) AuBr4-(aq)
B) Mn2+(aq)
C) K+(aq)
D) F2O(aq)
E) H+(aq)
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following solids is commonly used as an inactive electrode in electrochemical cells?
A) zinc
B) graphite
C) copper
D) iron
E) sodium
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A cell can be prepared from copper and tin. What is the E°cell for the cell that forms from the following half-reactions?
Cu2+(aq) + 2e-  Cu(s) E° = 0.34 V
Sn4+(aq) + 2e-
Cu(s) E° = 0.34 V
Sn4+(aq) + 2e-  Sn2+(aq) E° = 0.13 V
Sn2+(aq) E° = 0.13 V
A) 0.47 V
B) 0.21 V
C) -0.21 V
D) -0.47 V
E) 0.42 V
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A current of 250. A flows for 24.0 hours at an anode where the reaction occurring is Mn2+(aq) + 2H2O(l) MnO2(s) + 4H+(aq) + 2e- What mass of MnO2 is deposited at this anode?
A) 19.5 kg
B) 12.9 kg
C) 4.87 kg
D) 2.43 kg
E) none of the above
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A voltaic cell consists of an Au/Au3+ electrode (E° = 1.50 V) and a Cu/Cu2+ electrode (E° = 0.34 V) . Calculate [Au3+] if [Cu2+] = 1.20 M and Ecell = 1.13 V at 25°C.
A) 0.001 M
B) 0.002 M
C) 0.01 M
D) 0.02 M
E) 0.04 M
G) B) and C)
Correct Answer

verified
Correct Answer
verified
True/False
A buried iron pipe can be protected against corrosion by connecting it to a rod of magnesium.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When the following redox equation is balanced with smallest whole number coefficients, the coefficient for Sn(OH) 3- will be _____. Bi(OH) 3(s) + Sn(OH) 3-(aq) Sn(OH) 62-(aq) + Bi(s) (basic solution)
A) 1
B) 2
C) 3
D) 6
E) none of the above
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the reaction of iodine with manganese dioxide
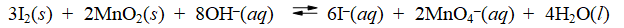 The equilibrium constant for the overall reaction is 8.30 × 10-7. Calculate G° for the reaction at 25°C.
The equilibrium constant for the overall reaction is 8.30 × 10-7. Calculate G° for the reaction at 25°C.
A) -15.1 kJ
B) -34.7 kJ
C) 15.1 kJ
D) 34.7 kJ
E) none of the above
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following statements relating to the glass electrode is correct?
A) The glass electrode detects hydrogen gas.
B) The glass of a glass electrode serves to conduct electrons.
C) When pH is measured, only a single electrode, the glass electrode, need be used.
D) The potential of the glass electrode varies linearly with the pH of the solution.
E) None of the above statements is correct.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 102
Related Exams