A) H2O
B) CN-
C) NH3
D) OH-
E) Cl-
G) A) and D)
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
Consider the following octahedral complex structures,each involving ethylene diamine and two different,unidentate ligands X and Y. 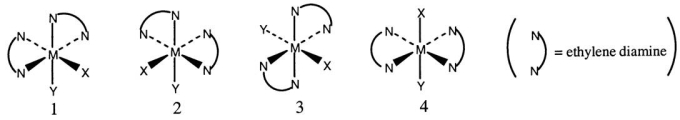 Which one of the following statements about these structures is incorrect?
Which one of the following statements about these structures is incorrect?
A) Structures 1 and 2 are optical isomers.
B) Structures 1 and 3 are optical isomers.
C) Structures 1 and 3 are different complexes.
D) Structures 1 and 4 are geometrical isomers.
E) Structures 3 and 4 are the same complex.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following will be the strongest oxidizing agent?
A) Cr
B) Cr(II)
C) Cr(III)
D) Cr(IV)
E) Cr(VI)
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The crystal field splitting energy, ,
A) is larger for tetrahedral complexes than for octahedral complexes.
B) depends on the metal but not on the ligand.
C) determines the color of a complex.
D) is larger for ionic ligands like chloride than for molecular ligands like carbon monoxide,CO.
E) determines the charge of a complex.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to valence bond theory,what would be the set of hybrid orbitals used when a Period 4 transition metal forms a tetrahedral complex?
A) d2sp
B) dsp2
C) dsp3
D) sp3
E) d2p2
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the coordination number of cobalt in the complex ion [Co(en) Cl4]-? (en = ethylenediamine)
A) 1
B) 2
C) 4
D) 6
E) 8
G) A) and D)
Correct Answer

verified
Correct Answer
verified
True/False
Tetrahedral complexes can exhibit both optical and linkage isomerism.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Write the formula for sodium tetracyanonickelate(II) .
A) Na[Ni(CN) 4]
B) Na[Ni(CN) 4]2
C) Na2[Ni(CN) 4]
D) Na4[Ni(CN) 4]
E) None of these choices is correct.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Essay
Apply the valence bond theory to predict the electronic structure and hybridization pattern of chromium in the complex ion Cr(NH3)63+.
Correct Answer

verified
The three d electrons of the Cr(III)will occupy three 3d orbitals,singly.The remaining two 3d orbitals will be hybridized with the 4s and 4p orbitals,yielding six d2sp3 hybrid orbitals,which are used to accommodate the six lone pairs of the ammonia ligands.
Correct Answer
verified
Multiple Choice
If M represents a transition element,which of the following oxides should be the least basic?
A) MO
B) M2O
C) M2O3
D) MO2
E) MO3
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the compound [Ni(en) 2(H2O) 2]SO4 (where en = ethylenediamine) the oxidation number and coordination number of nickel are,respectively:
A) 2 and 6
B) 4 and 6
C) 6 and 6
D) 2 and 4
E) 4 and 4
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following transition elements will form an ion with the largest oxidation number?
A) chromium,Cr,Group 6B(6)
B) manganese,Mn,Group 7B(7)
C) iron,Fe,Group 8B(8)
D) cobalt,Co,Group 8B(9)
E) zinc,Zn,Group 2B(12)
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Essay
The compound Rh(CO)(H)(PH3)2 forms cis and trans isomers.Use this information to predict the geometry of this complex,and draw the geometric isomers.
Correct Answer

verified
The complex has four ligands a...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
In a coordination compound involving a complex ion of square planar geometry,which of the following types of isomerism is/are never possible?
A) geometric
B) optical
C) linkage
D) coordination
E) more than one of the above
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following ions could exist in only the high-spin state in an octahedral complex?
A) Cr2+
B) Mn4+
C) Fe3+
D) Co3+
E) Ni2+
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The ground state electron configuration of Cr2+ is
A) [Ar]4s13d5
B) [Ar]4s23d4
C) [Ar]3d4
D) [Ar]4s13d3
E) [Ar]4s23d2
G) A) and D)
Correct Answer

verified
C
Correct Answer
verified
True/False
Of the 3d transition series of elements,scandium has the greatest atomic radius.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The oxidation and coordination numbers of cobalt in the compound [Co(NH3) 5Cl]Cl2 are,respectively:
A) 2 and 6
B) 2 and 8
C) 3 and 6
D) 3 and 8
E) None of these choices is correct.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following species could exist as isomers?
A) [Co(H2O) 4Cl2]+
B) [Pt(NH3) Br3]-
C) [Pt(en) Cl2]
D) [Pt(NH3) 3Cl]+
E) None of these choices is correct.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
What geometry is particularly common for complexes of d10 metal ions?
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 72
Related Exams