A) 0.5 m C12H22O11 (sucrose)
B) 0.5 m Ca(NO3) 2
C) 0.5 m NiSO4
D) 0.5 m Li3PO4
E) pure water
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The solubility of the oxidizing agent potassium permanganate is 7.1 g per 100.0 g of water at 25°C.What is the mole fraction of potassium permanganate in this solution?
A) 0.0080
B) 0.0086
C) 0.066
D) 0.45
E) 0.48
G) All of the above
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
Select the weakest electrolyte from the following set.
A) Na2SO4
B) KCl
C) CH3CH2COOH,propionic acid
D) CaCl2
E) LiOH
G) A) and C)
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
Calculate the freezing point of a solution made by dissolving 3.50 g of potassium chloride ( 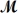 = 74.55 g/mol) in 100.0 g of water.Assume ideal behavior for the solution;Kf = 1.86°C/m.
= 74.55 g/mol) in 100.0 g of water.Assume ideal behavior for the solution;Kf = 1.86°C/m.
A) -1.7°C
B) -0.9°C
C) 0.0°C
D) 0.9°C
E) 1.7°C
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
A 50.0 % (w/w)solution of sulfuric acid (H2SO4)in water has a density of 1.395 g/mL.What is its molarity?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Sodium hydroxide is a common ingredient in drain cleaners such as Drano.The mole fraction of sodium hydroxide in a saturated aqueous solution is 0.310.What is the molality of the solution?
A) 0.310 m
B) 0.690 m
C) 1.24 m
D) 12.4 m
E) 25.0 m
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
From the following list of aqueous solutions and water,select the one with the lowest freezing point.
A) 0.75 M (NH4) 3PO4
B) l.0 M CaSO4
C) l 0 M LiClO4
D) 1.5 M CH3OH,methyl alcohol
E) pure water
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 0.89% (w/v) sodium chloride solution is referred to as physiological saline solution because it has the same concentration of salts as human blood.What is the molarity of a physiological saline solution?
A) 0.0028 M
B) 0.015 M
C) 0.15 M
D) 0.30 M
E) 0.35 M
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Raoult's Law relates the vapor pressure of the solvent above the solution to its mole fraction in the solution.Which of the following is an accurate statement?
A) Raoult's Law applies exactly to all solutions.
B) Raoult's Law works best when applied to concentrated solutions.
C) Raoult's Law works best when applied to dilute solutions.
D) Raoult's Law applies only to non-ideal solutions.
E) None of these statements is accurate.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the strongest electrolyte from the following set.
A) CH3CH2OH,ethanol
B) LiNO3
C) C6H12O6,glucose
D) CCl4
E) HF
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Methane has a Henry's Law constant (k) of 9.88 10-2 mol/(L·atm) when dissolved in benzene at 25°C.How many grams of CH4 will dissolve in 3.00 L of benzene if the partial pressure of CH4 is 1.48 atm?
A) 0.0667 g
B) 0.146 g
C) 2.34 g
D) 4.83 g
E) 7.02 g
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Saccharin,one of the first non-nutritive sweeteners used in soft-drinks,is 500 times sweeter than sugar in dilute aqueous solutions.The solubility of saccharin is 1.00 gram per 290 mL of solution.What is the molarity of a saturated saccharin solution?  saccharin= 183.2 g/mol
saccharin= 183.2 g/mol
A) 0.0188 M
B) 0.632 M
C) 1.58 M
D) 3.45 M
E) None of these choices is correct.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following aqueous solutions should demonstrate the most ideal behavior?
A) 0.1 M K2SO4
B) 0.1 M CaCl2
C) 3.0 M LiF
D) 0.1 M MgSO4
E) 0.1 M NaCl
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
The density of pure water at 25°C is 0.997 g/mL.Considering water as being both solvent and solute,calculate its molarity.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The mole fraction of potassium nitrate in an aqueous solution is 0.0194.The solution's density is 1.0627 g/mL.Calculate the molarity of the solution.
A) 0.0194 M
B) 0.981 M
C) 1.05 M
D) 1.96 M
E) 19.4 M
G) A) and C)
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
Which of the following statements describes the correct method of preparation of 1.00 L of a 2.0 M urea solution? 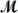 urea = 60.06 g/mol
urea = 60.06 g/mol
A) Dissolve 120 g of urea in 1.00 kg of distilled water.
B) Dissolve 120 g of urea in 880 g of distilled water.
C) Dissolve 120 g of urea in enough distilled water to produce 1.00 L of solution.
D) Dissolve 120 g of urea in 1.00 liter of distilled water.
E) The density of urea is needed in order to do this calculation.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For a given solution,which of the following concentration values will change as temperature changes?
A) mass percent
B) molality
C) mole fraction
D) molarity
E) None of these choices is correct.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
True/False
The solubility of gases in water increases with increase in the pressure of the gas.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The chemist,Anna Lytic,must prepare 1.00 kg of 15.0% (w/w) acetic acid using a stock solution which is 36.0% (w/w) acetic acid (d = 1.045 g/mL) .Which of the following combinations will give her the solution she wants?
A) 417 mL of 36% acetic acid in 583 mL of distilled water
B) 417 g of 36% acetic acid in 583 g of distilled water
C) 360 mL of 36% acetic acid in 640 mL of distilled water
D) 360 g of 36% acetic acid in 640 g of distilled water
E) 150 g of 36% acetic acid in 850 g of distilled water
G) All of the above
Correct Answer

verified
Correct Answer
verified
True/False
The heat of solution is the total enthalpy change when a solution is formed from the separated solute and solvent.
B) False
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 73
Related Exams