A) [Ne]3s13p1
B) [He]2s12p3
C) [Ne]3s23p23d1
D) [Ne]3s23p33d1
E) [Ne]3s23p3
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following elements has the smallest atomic radius?
A) Li
B) Ne
C) Rb
D) Sr
E) Xe
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following elements has the greatest atomic radius?
A) Li
B) Ne
C) Rb
D) Sr
E) Xe
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The electronic structure 1s22s22p63s23p64s23d8 refers to the ground state of
A) Kr
B) Ni
C) Fe
D) Pd
E) None of these choices is correct.
G) All of the above
Correct Answer

verified
Correct Answer
verified
True/False
According to the exclusion principle,two is the maximum number of electrons in an atom which can share the same four quantum numbers.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the correct electron configuration for Te (Z = 52) .
A) [Kr]5s25p64d8
B) [Kr]5s25d105p4
C) [Kr]5s24d105p6
D) [Kr]5s24f14
E) [Kr]5s24d105p4
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the correct set of quantum numbers (n,l,ml,ms) for the highest energy electron in the ground state of potassium,K.
A) 4,1,-1,![]()
B) 4,1,0,![]()
C) 4,0,1,![]()
D) 4,0,0,![]()
E) 4,1,1,![]()
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the element with the most negative electron affinity (i.e. ,accepts an electron most readily) .
A) H
B) Li
C) C
D) F
E) Ne
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following equations correctly represents the process involved in the electron affinity of X?
A) X(g) X+(g) + e-
B) X+(g) X+(aq)
C) X+(g) + e- X(g)
D) X(g) + e- X-(g)
E) X+(g) + Y-(g) XY(s)
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the correct electron configuration for Cu (Z = 29) .
A) [Ar]4s23d9
B) [Ar]4s13d10
C) [Ar]4s24p63d3
D) [Ar]4s24d9
E) [Ar]5s24d9
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following statements about orbital energies is incorrect?
A) In the hydrogen atom,the energy of an orbital depends only on the value of the quantum number n.
B) In many-electron atoms the energy of an orbital depends on both n and l.
C) Inner electrons shield outer electrons more effectively than do electrons in the same orbital.
D) The splitting of sublevels in many-electron atoms is explained in terms of the penetration effect.
E) The energy of a given orbital increases as the nuclear charge Z increases.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The most acidic oxides are formed from elements found in the _________________ region of the periodic table.
A) upper right
B) upper left
C) center
D) lower right
E) lower left
G) B) and D)
Correct Answer

verified
Correct Answer
verified
True/False
The difference in energies between the 1s and 2s orbitals is due to the penetration effect.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following equations correctly represents the process relating to the ionization energy of X?
A) X(s) X+(g) + e-
B) X2(g) X+(g) + X-(g)
C) X(g) + e- X-(g)
D) X-(g) X(g) + e-
E) X(g) X+(g) + e-
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An atom of element number 33 (As) is in its ground electronic state.Which one of the following sets of quantum numbers could not apply to any of its electrons? 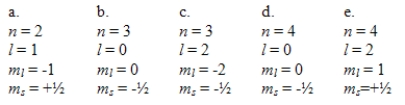
A) a
B) b
C) c
D) d
E) e
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Elements with _______________ first ionization energies and ___________ electron affinities generally form anions.
A) low,very negative
B) high,positive or slightly negative
C) low,positive or slightly negative
D) high,very negative
E) None of these is generally correct.
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In a single atom,what is the maximum number of electrons which can have quantum number n = 4?
A) 16
B) 18
C) 32
D) 36
E) None of these choices is correct.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
True/False
Elements in which the outermost electron has the same principal quantum number n,show similar chemical properties.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The ____________________ quantum numbers are associated with the energy of an electron in a many-electron atom.
A) n and l
B) n and ml
C) l and ml
D) n and ms
E) n,l and ml
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following statements about atomic structure and quantum numbers is incorrect?
A) In a given atom,the maximum number of electrons having principal quantum number n = 3,is 18.
B) The number of orbitals in a given f subshell is 7.
C) For n = 4,the largest possible value of l is 3.
D) For n = 4,the largest possible value of ml is 2.
E) The following set of quantum numbers for a single orbital is not allowed: n = 3,l = 1,ml = -2.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 70
Related Exams