A) 2.2 × 10-13
B) 2.2 × 10-12
C) 2.9 × 10-13
D) 0.050
E) 4.5 × 10-13
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Ammonia is a ________.
A) weak acid
B) strong base
C) weak base
D) strong acid
E) salt
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The 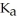 of hypochlorous acid (HClO) is 3.0 ×
of hypochlorous acid (HClO) is 3.0 × 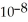 at 25.0 °C.Calculate the pH of a
at 25.0 °C.Calculate the pH of a 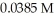 hypochlorous acid solution.
hypochlorous acid solution.
A) 3.05
B) 9.53
C) 4.47
D) 6.52
E) -3.05
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Of the following,which is the strongest acid?
A) HClO
B) HClO3
C) HClO2
D) HClO4
E) HIO
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following 0.5 M aqueous salt solutions will have a pH of 7.0 at 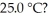 LiF RbBr NaClO4 NH4Cl
LiF RbBr NaClO4 NH4Cl
A) LiF only
B) NaClO4 only
C) LiF and RbBr
D) RbBr and NaClO4
E) NH4Cl only
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The pH of an aqueous solution at 25.0 °C is 10.55.What is the molarity of 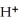 in this solution?
in this solution?
A) 2.8 × 10-11
B) 3.5 × 10-4
C) 3.45
D) 1.1 × 10-13
E) 3.5 × 1010
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Of the following,which is the weakest acid?
A) HPO3-
B) H3PO4
C) H2PO4-
D) HPO4-
E) The acid strength of all of the above is the same.
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The Ka for HCN is 4.9 × 10-10.What is the value of Kb for CN-?
A) 2.0 × 10-5
B) 4.0 × 10-6
C) 4.9 × 104
D) 4.9 × 10-24
E) 2.0 × 109
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Using the data in the table,which of the conjugate bases below is the weakest base? 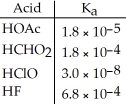
A) OAc-
B) CHO2-
C) ClO-
D) F-
E) OAc- and CHO2-
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The hydride ion, 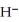 ,is a stronger base than the hydroxide ion,O
,is a stronger base than the hydroxide ion,O 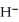 .The product(s) of the reaction of hydride ion with water is/are ________.
.The product(s) of the reaction of hydride ion with water is/are ________.
A) H3O+ (aq)
B) OH- (aq) + H2 (g)
C) OH- (aq) + 2H+ (aq)
D) no reaction occurs
E) H2O2 (aq)
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The acid-dissociation constants of sulfurous acid (H2SO3) are Ka1 = 1.7 × 10-2 and 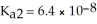 at 25.0 °C.Calculate the pH of a 0.163 M aqueous solution of sulfurous acid.
at 25.0 °C.Calculate the pH of a 0.163 M aqueous solution of sulfurous acid.
A) 4.53
B) 1.28
C) 1.86
D) 6.21
E) 1.93
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The 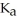 for acid HA is
for acid HA is 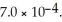 What is the pH of a 0.15 M aqueous solution of KA?
What is the pH of a 0.15 M aqueous solution of KA?
A) 0.82
B) 8.17
C) 5.83
D) 1.17
E) 5.01
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the pOH of a 0.030 M solution of calcium hydroxide?
A) 12.78
B) 1.22
C) 12.48
D) 10.41
E) 1.52
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following is the weakest acid?
A) HF (Ka = 6.8 × 10-4)
B) HClO (Ka = 3.0 × 10-8)
C) HNO2 (Ka = 4.5 × 10-4)
D) HCN (Ka = 4.9 × 10-10)
E) Acetic acid (Ka = 1.8 × 10-5)
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Using the data in the table,which of the conjugate bases below is the weakest base? 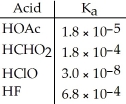
A) OAc-
B) CHO2-
C) ClO-
D) F-
E) OAc- and CHO2-
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The conjugate base of NH3 is ________.
A) NH2-
B) NH4+
C) NH2OH
D) H3O+
E) OH-
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The 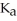 of citric acid is 1.9 ×
of citric acid is 1.9 × 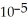 at 25.0 °C.What is the pH of a 0.35 M aqueous solution of citric acid?
at 25.0 °C.What is the pH of a 0.35 M aqueous solution of citric acid?
A) 0.46
B) 2.59
C) 5.23
D) -3.46
E) 2.13
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Hydrochloric acid is a strong acid.This means that ________.
A) HCl dissociates completely to H+(aq) and Cl-(aq) when it dissolves in water
B) HCl does not dissociate at all when it is dissolved in water
C) HCl produces a gaseous product when it is neutralized
D) HCl cannot be neutralized by a weak base
E) aqueous solutions of HCl contain equal concentrations of H+(aq) and OH-(aq)
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An aqueous solution contains 0.390 M HCl at 25.0 °C.The pH of the solution is ________.
A) 0.41
B) 0.390
C) 13.61
D) 13.59
E) 7.00
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Ka for arsenic acic,HAsO42-,is 7.5 × 10-12.What is the pH of a 0.15 M aqueous solution of AsO43-?
A) 7.97
B) 12.10
C) 8.04
D) 1.96
E) 6.00
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 139
Related Exams