A) DNA polymerase helps synthesize new DNA before our cells divide.
B) Antibodies regulate sweat to keep infections out of our skin pores.
C) Collagen is a structural protein to support hair, skin, and nails.
D) Insulin regulates blood glucose levels.
E) Hemoglobin protein transports oxygen to our cells.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In an ionic bond,
A) two atoms both become strongly electronegative and attract each other.
B) atoms attract each other by sharing electrons to fill their valence shells.
C) atoms, having gained or lost electrons, attract one another with opposite charges.
D) two atoms are attracted by partial positive and negative charges.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Figuer:
Examine this image of the glucose molecule. 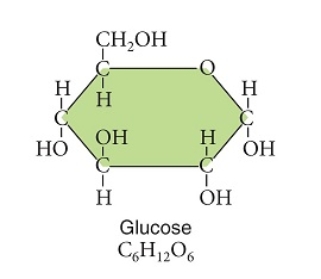 -Compared with a molecule of glucose,this starch molecule does NOT have which characteristic below?
-Compared with a molecule of glucose,this starch molecule does NOT have which characteristic below? 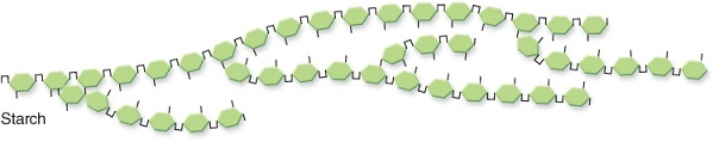
A) This molecule is used by cells for long-term storage and release of energy for cell functions.
B) This molecule is used by cells for quick release of energy for cell functions.
C) This molecule can provide structure for cells that contain it.
D) This molecule is a complex carbohydrate polymer.
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The unique properties of water,including its strength as a solvent,its three environmental stages of solid,liquid,and gas,and its temperature regulation,are a result of
A) symmetric balance of electronegativity as shared electrons orbit equally around the hydrogens and oxygens.
B) the cohesion and adhesion of water molecules that bond more strongly to each other than other substances.
C) unbalanced electronegativity of the hydrogens and oxygens as they share electrons.
D) the imbalance in numbers of electrons around hydrogen and oxygen valence shells after they ionically bond.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
True/False
Proteins store the genetic information of the cell and transmit it to the next generation.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which statement summarizes the distinction between nonpolar and polar covalent bonds?
A) The electrons are more evenly and symmetrically distributed in orbit among atoms in a nonpolar covalent bond.
B) Polar covalent bonds are formed when the atoms gain or lose electrons to bond, and become oppositely charged ions.
C) The difference in electronegativity of the atoms in a nonpolar covalent bond is very large.
D) The electrons are more evenly and symmetrically distributed in orbit among atoms in a polar covalent bond.
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A base
A) is a chemical that absorbs hydrogen ions from a solution.
B) has a value of 7 on the pH scale.
C) has a value below 7 on the pH scale.
D) is a chemical that adds hydrogen ions to a solution.
F) None of the above
Correct Answer

verified
Correct Answer
verified
True/False
A peptide bond is a covalent bond formed between the amino group of one amino acid and the R group of another amino acid.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The mass number is defined as the total number of __________ of an atom.
A) neutrons and electrons
B) protons
C) protons, neutrons, and electrons
D) protons and neutrons
E) protons and electrons
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In a covalent bond,atoms
A) share a proton.
B) share electrons.
C) of opposite charges attract each other.
D) both become highly electronegative.
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The primary elements making up living organisms are
A) carbon, hydrogen, oxygen, sulfur, nitrogen, and phosphorus.
B) carbon, hydrogen, iron, sulfur, sodium, and calcium.
C) carbon, oxygen, iron, chlorine, sulfur, and phosphorus.
D) carbon, hydrogen, oxygen, calcium, iron, and iodine.
E) carbon, oxygen, sulfur, calcium, iron, and phosphorus.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An acid
A) has a value above seven on the pH scale.
B) is a chemical that takes hydrogen ions from a solution.
C) has a value of seven on the pH scale.
D) is a chemical that adds hydrogen ions to a solution.
E) All of the answer choices are correct.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The four nitrogen bases found in RNA are
A) adenine, thymine, guanine, and uracil.
B) adenine, cytosine, guanine, and uracil.
C) adenine, thymine, cytosine, and uracil.
D) thymine, cytosine, guanine, and uracil.
E) None of the answer choices are correct.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A conscientious person habitually reads nutrition labels on food packages for weight watching and general health.The main nutritional molecules are made up of
A) buffers.
B) trace elements.
C) isotopes.
D) bulk elements.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
True/False
Figuer:
Examine these two sugars, as shown prior to the chemical reaction that would bond them. 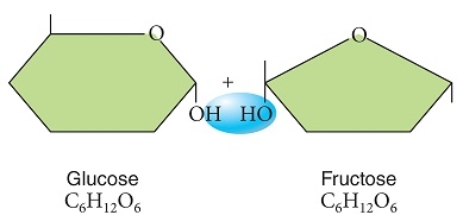 -These glucose and fructose molecules will bond to form a monosaccharide with the removal of water.
-These glucose and fructose molecules will bond to form a monosaccharide with the removal of water.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Evaporation of water is
A) a phase change of water from liquid into a vapor.
B) a phase change of water from solid into a vapor.
C) a phase change of water from vapor into a liquid.
D) a phase change of water from vapor into a solid.
E) All of the answer choices are correct.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The atomic number of an element is the number of
A) protons in the orbitals.
B) neutrons in the orbitals.
C) neutrons in the nucleus.
D) protons in the nucleus.
E) electrons in the nucleus.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
True/False
The property of water demonstrated by this water strider,as it remains on top of the water,is that water is a universal solvent. 
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An element is found to have atoms with eight electrons in its valence shell.The atoms will be ____
A) highly reactive.
B) not chemically stable.
C) chemically stable.
D) highly likely to combine with other atoms.
E) not inert.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Saturated fats have long straight tails of fatty acids,while unsaturated fats from vegetables have kinks in their tails due to double bonds.These kinks prevent the fats from packing together as tightly.Hydrogenated vegetable oils,or trans fats,have hydrogens added back to the double bonds and thus behave like
A) carbohydrates.
B) waxes.
C) unsaturated fats.
D) saturated fats.
E) proteins.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 68
Related Exams