A) Is highly reactive
B) Is not chemically stable
C) Is highly likely to combine with other atoms
D) Is not inert
E) Is chemically stable
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Examples of monosaccharides are:
A) Glucose, maltose, and cellulose
B) Glucose, lactose, and maltose
C) Glucose, galactose, and fructose
D) Glucose, lactose, and cellulose
E) None of these are correct
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Evaporation is:
A) The conversion of a liquid into a vapor
B) The conversion of a solid into a vapor
C) The conversion of a vapor into a liquid
D) The conversion of a vapor into a solid
E) All are correct
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The most common isotope of carbon is 12C.14C will have ____ than 12C.
A) More protons
B) More neutrons
C) Fewer neutrons
D) Fewer protons
E) More electrons
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In Table 2.7 why were amino acids analyzed for 15N? 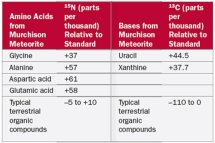
A) Nitrogen is not present in nucleotides or carbohydrates
B) Nitrogen is not present in nucleotides or lipids
C) Nitrogen is not present in carbohydrates or lipids
D) Nitrogen is not present in amino acids or carbohydrates
E) Nitrogen is not present in nucleotides or amino acids
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The primary elements making up living organisms are:
A) Carbon, hydrogen, oxygen, calcium, iron, and iodine
B) Carbon, oxygen, iron, chlorine, sulfur, and phosphorous
C) Carbon, hydrogen, iron, sulfur, sodium, and calcium
D) Carbon, hydrogen, oxygen, sulfur, nitrogen, and phosphorous
E) Carbon, oxygen, sulfur, calcium, iron, and phosphorous
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Isotopes of the same element are different from one another in that:
A) They have a different number of protons
B) They have a different number of neutrons
C) They have a different number of electrons
E) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The polymers with the most complex and diverse three-dimensional structure are:
A) Saturated fats
B) Unsaturated fats
C) Proteins
D) Waxes
E) Carbohydrates
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A substance having a pH of 6 would best be described as:
A) A weak acid
B) Neutral
C) A weak base
D) A strong acid
E) A strong base
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What essential function would lipids play in the origin of life?
A) The formation of membranes
B) Storage of information
C) Catalysis of reactions
D) Formation of a cytoskeleton
E) Anaerobic fermentation
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The four nitrogen bases found in RNA are:
A) Adenine, thymine, guanine, and uracil
B) Adenine, cytosine, guanine, and uracil
C) Adenine, thymine, cytosine, and uracil
D) Thymine, cytosine, guanine, and uracil
E) None of these are correct
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
14C and 14N are both:
A) Atoms
B) Molecules
C) Compounds
D) Polymers
E) Ions
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What hypothesis were the scientists testing?
A) Can organic molecules be made on Earth?
B) Can extraterrestrial life be detected on meteorites?
C) Can life be created from a mixture of organic molecules?
D) What conditions existed on Earth when life first began?
E) Were organic molecules found on meteorites extraterrestrial?
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How are the monomers in proteins joined?
A) Phosphodiester bonds between amino acids
B) Peptide bonds between amino acids
C) Peptide bonds between nucleotides
D) Phosphodiester bonds between nucleotides
E) Peptide bonds between carbohydrates
G) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
Cohesion is a property of water in which water molecules tend to stick together.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following conclusions can be made from Table 2.7? 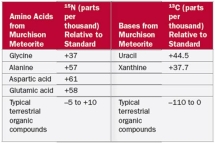
A) Glycine is a more abundant amino acid in the meteorite than in terrestrial samples
B) 15N is more abundant in amino acids from the meteorite than from terrestrial samples
C) Amino acids in the meteorite contain more nitrogen than amino acids from terrestrial samples
D) 13C is more abundant in amino acids from the meteorite than from terrestrial samples
E) Uracil is a more abundant amino acid in the meteorite than in terrestrial samples
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Trees take up water hundreds of feet away by:
A) Water's high density
B) Cohesion
C) Water's high boiling point
D) Adhesion
E) A neutral pH
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the example of ionic bond formation between sodium and chlorine:
A) Na is the chemical symbol for chlorine
B) Sodium accepts an electron
C) Chlorine accepts an electron
D) Chlorine becomes positively charged
E) Both sodium accepts an electron and chlorine becomes positively charged are correct
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A substance having a pH of 13 would best be described as:
A) A weak acid
B) A weak base
C) Neutral
D) A strong acid
E) A strong base
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A nucleotide contains which of the following?
A) Amino acid
B) Sugar
C) Sugar and nitrogenous base
D) Nitrogenous base
E) Nitrogenous base and amino acid
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 76
Related Exams