A) 658 K
B) 683 K
C) 955 K
D) 1047 K
E) 1229 K
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Elemental boron can be formed by the reaction of boron trichloride with hydrogen. 2BCl3(g) + 3H2(g) → 2B(s) + 6HCl(g)
What is ΔG°rxn for this reaction? 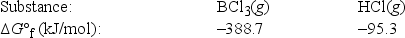
A) -293.4 kJ/mol
B) 293.4 kJ/mol
C) -205.6 kJ/mol
D) 205.6 kJ/mol
E) -2141.6 kJ/mol
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is ΔS° for the reaction SO2(s) + NO2(g) → SO3(g) + NO(g) ? 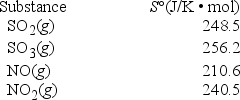
A) 53.6 J/K • mol
B) -53.6 J/K • mol
C) -22.2 J/K • mol
D) 474.8 J/K • mol
E) -474.8 J/K • mol
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is ΔG°rxn for the following reaction? SiCl4(g) + 2Mg(s) → 2MgCl2(s) + Si(s) 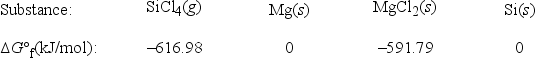
A) 566.60 kJ/mol
B) 50.38 kJ/mol
C) 25.19 kJ/mol
D) -25.19 kJ/mol
E) -566.60 kJ/mol
G) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
In a spontaneous process, the entropy of the system always increases.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The temperature at which the following process reaches equilibrium at 1.0 atm is the normal melting point for phosphoric acid. H3PO4(s)  H3PO4(l)
Use the following thermodynamic data at 298 K to estimate this temperature.
H3PO4(l)
Use the following thermodynamic data at 298 K to estimate this temperature. 
A) 286 K
B) 305 K
C) 315 K
D) 347 K
E) 3170 K
G) All of the above
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
Elemental boron can be formed by the reaction of boron trichloride with hydrogen. 2BCl3(g) + 3H2(g) → 2B(s) + 6HCl(g) 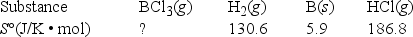
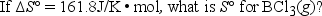
A) -18.2 J/K • mol
B) 18.2 J/K • mol
C) 289.5 J/K • mol
D) 370.4 J/K • mol
E) 579.0 J/K • mol
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many types of vibrations does a diatomic molecule exhibit?
A) 0
B) 1
C) 2
D) 3
E) 4
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is ΔS° for the reaction SO3(g) + H2O(l) → H2SO4(aq) ? 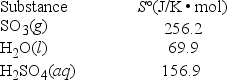
A) 169.2 J/K • mol
B) 343.2 J/K • mol
C) -169.2 J/K • mol
D) -29.4 J/K • mol
E) 29.4 J/K • mol
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Is the following process spontaneous? 
A) Yes, because ΔH > 0.
B) No, because ΔH < 0.
C) Yes, because ΔS > 0.
D) No, because ΔS < 0.
E) It's impossible to tell without knowing the temperature of the system.
G) A) and B)
Correct Answer

verified
E
Correct Answer
verified
Multiple Choice
Consider the figure below which shows ΔG° for a chemical process plotted against absolute temperature. From this plot, it is reasonable to conclude that: 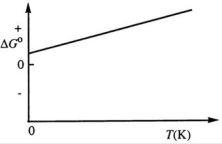
A) ΔH° > 0, ΔS° > 0
B) ΔH° > 0, ΔS° < 0
C) ΔH° < 0, ΔS° > 0
D) ΔH° < 0, ΔS° < 0
E) None of these choices is correct.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the figure below which shows ΔG° for a chemical process plotted against absolute temperature. From this plot, it is reasonable to conclude that: 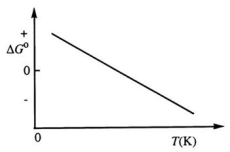
A) ΔH° > 0, ΔS° > 0
B) ΔH° > 0, ΔS° < 0
C) ΔH° < 0, ΔS° > 0
D) ΔH° < 0, ΔS° < 0
E) None of these choices is correct.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which statement is correct?
A) Reaction of ADP and alanine results in ATP in the body.
B) A positive ΔG° value means that the reaction is not product favored.
C) CO2 is released when ADP reacts with an amino acid.
D) Phosphoric acid is one of the products of the reaction of ADP with alanine.
E) Hydrolysis of ATP requires NaOH.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
Which thermodynamic property is a measure of the disorder in the system?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is defined as a process that may occur under a specific set of conditions?
A) Conditional process
B) Nonspontaneous process
C) Specified process
D) Spontaneous process
E) Conditionality law
G) A) and E)
Correct Answer

verified
D
Correct Answer
verified
Multiple Choice
What is ΔS° at 25°C for the reduction of lead(II) oxide by charcoal? 2PbO(s) + C(s) → 2Pb(s) + CO2(g) 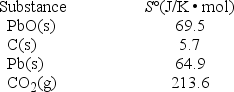
A) +198.7 J/K • mol
B) +488.0 J/K • mol
C) +353.7J/K • mol
D) -203.3 J/K • mol
E) +203.3 J/K • mol
G) None of the above
Correct Answer

verified
Correct Answer
verified
Short Answer
How does the entropy change when a solid is melted?
Correct Answer

verified
Correct Answer
verified
Short Answer
If an endothermic process is spontaneous when |TΔS| >|ΔH|, then is ΔS is positive, negative, or equal to zero?
Correct Answer

verified
Correct Answer
verified
Short Answer
The ________ law of thermodynamics states that the entropy of the universe in a spontaneous process increases and is constant in equilibrium processes.
Correct Answer

verified
Correct Answer
verified
True/False
The entropy of vaporization of a compound is always positive.
B) False
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 107
Related Exams