A) Melting point
B) Triple point
C) Critical point
D) Sublimation point
E) Boiling point
G) A) and D)
Correct Answer

verified
Correct Answer
verified
True/False
Hexagonal close packing of identical atoms occurs when close-packed layers are stacked in an abcabc... arrangement.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
The phase diagram of a substance shows the energy changes associated with changes of state.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
Ceramic superconductors often contain copper in unusual oxidation states.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following should have the highest surface tension at a given temperature?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about ceramics is incorrect?
A) Silicon carbide has a diamond-like structure.
B) Boron nitride can exist in both diamond-like and graphite-like forms.
C) Silicon carbide can be prepared by direct reaction of silicon and carbon.
D) Superconducting ceramics present manufacturing difficulties owing to their brittleness.
E) Superconducting ceramic compounds usually incorporate cobalt in a key role.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What types of forces exist between molecules of CO2?
A) Hydrogen bonding only.
B) Hydrogen bonding and dispersion forces.
C) Dipole-dipole forces only.
D) Dipole-dipole and dispersion forces.
E) Dispersion forces only.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When the electron cloud of a molecule is easily distorted, the molecule has a high
A) polarity.
B) polarizability.
C) dipole moment.
D) van der Waals radius.
E) compressibility.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Lead crystallizes in the face-centered cubic lattice. What is the coordination number for Pb?
A) 4
B) 6
C) 8
D) 10
E) 12
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In which of the following compounds will the molecules not form hydrogen bonds with each other?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) C) and D)
Correct Answer

verified
Correct Answer
verified
True/False
Liquid crystal displays are most commonly constructed of smectic type liquid crystals.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A metal with a body-centered cubic lattice will have __________ atom(s) per unit cell.
A) 1
B) 2
C) 3
D) 4
E) 9
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Examine the following phase diagram and identify the feature represented by point A. 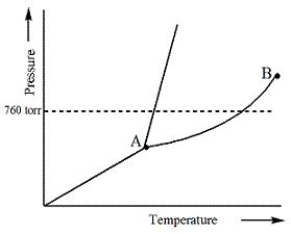
A) Melting point
B) Critical point
C) Triple point
D) Sublimation point
E) Boiling point
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The phase diagram for xenon has a solid-liquid curve with a positive slope. Which of the following is true?
A) Solid xenon has a higher density than liquid xenon.
B) Solid xenon has the same density as liquid xenon.
C) The phase diagram cannot be used to predict which phase of xenon is denser.
D) Freezing xenon is an endothermic process.
E) None of these choices are correct.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the pair of substances in which the one with the higher vapor pressure at a given temperature is listed first.
A) C 7H 16, C 5H 12
B) CCl 4, CBr 4
C) H 2O, H 2S
D) CH 3CH 2OH, CH 3-O-CH 3
E) Xe, Kr
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Diethyl ether, used as a solvent for extraction of organic compounds from aqueous solutions, has a high vapor pressure which makes it a potential fire hazard in laboratories in which it is used. How much energy is released when 100.0 g is cooled from 53.0°C to 10.0°C? 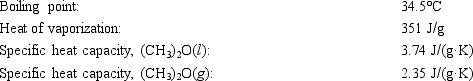
A) 10.1 kJ
B) 13.1 kJ
C) 16.1 kJ
D) 45.2 kJ
E) 48.6 kJ
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements concerning a face-centered cubic unit cell and the corresponding lattice, made up of identical atoms, is incorrect?
A) The coordination number of the atoms in the lattice is 8.
B) The packing in this lattice is more efficient than for a body-centered cubic system.
C) If the atoms have radius r, then the length of the cube edge is √8 × r.
D) There are four atoms per unit cell in this type of packing.
E) The packing efficiency in this lattice and hexagonal close packing are the same.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
True/False
A liquid may be made to boil at room temperature, simply by lowering the pressure.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Octane has a vapor pressure of 40. torr at 45.1°C and 400. torr at 104.0°C. What is its heat of vaporization?
A) 39.0 kJ/mol
B) 46.0 kJ/mol
C) 590 kJ/mol
D) 710 kJ/mol
E) None of these choices are correct.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Examine the phase diagram for the substance Bogusium (Bo) and select the correct statement. 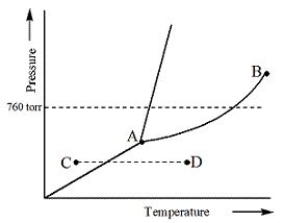
A) Bo( s) has a lower density than Bo( l) .
B) The triple point for Bo is at a higher temperature than the melting point for Bo.
C) Bo changes from a solid to a liquid as one follows the line from C to D.
D) Bo changes from a liquid to a gas as one follows the line from C to D.
E) Point B represents the critical temperature and pressure for Bo.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 95
Related Exams