A) CaS( s)
B) BaO( s)
C) NaI( s)
D) LiBr( s)
E) MgO( s)
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Using the bond energies provided below, calculate ΔH° for the reaction CH4(g) + 4Cl2(g) → CCl4(g) + 4HCl(g) Bond energies: C-H = 413 kJ/mol, Cl-Cl = 243 kJ/mol, C-Cl = 339 kJ/mol, H-Cl = 427 kJ/mol
A) 1422 kJ
B) 440 kJ
C) 110 kJ
D) −110 kJ
E) −440 kJ
G) A) and C)
Correct Answer

verified
E
Correct Answer
verified
Multiple Choice
In which of these substances are the atoms held together by metallic bonding?
A) CO 2
B) Si
C) Br 2
D) S 8
E) Cr
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
In covalent bond formation, the potential energy reaches a maximum when the internuclear distance is equal to the bond length.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds displays the greatest ionic character in its bonds?
A) NO 2
B) CO 2
C) H 2O
D) HF
E) NH 3
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following contains ionic bonding?
A) CO
B) SrF 2
C) Al
D) OCl 2
E) HCl
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The lattice energy for ionic crystals increases as the charge on the ions __________ and the size of the ions __________.
A) increases; increases
B) increases; decreases
C) decreases; increases
D) decreases; decreases
E) None of these choices are correct.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is an ionic compound?
A) H 2S
B) NH 3
C) I 2
D) KI
E) CCl 4
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following properties is least characteristic of typical metals?
A) Moderately high melting point
B) High boiling point
C) Brittleness
D) Good electrical conductor when solid
E) Good electrical conductor when molten
G) B) and C)
Correct Answer

verified
Correct Answer
verified
True/False
A single covalent bond consists of a single delocalized electron pair.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following properties is least characteristic of substances composed of small, covalently-bonded molecules?
A) Low melting point
B) Low boiling point
C) Weak bonds
D) Poor electrical conductor when solid
E) Poor electrical conductor when molten
G) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
The lattice energy of large ions is greater in magnitude than that of small ions of the same charge.
B) False
Correct Answer

verified
False
Correct Answer
verified
Multiple Choice
The lattice energy of CaF2 is the energy change for which one, if any, of the following processes?
A) Ca 2+( s) + 2F −( g) → CaF 2( g)
B) CaF 2( g) → CaF 2( s)
C) Ca( g) + 2F( g) → CaF 2( s)
D) CaF 2( aq) → CaF 2( s)
E) None of these choices are correct.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the most polar bond amongst the following.
A) C-O
B) Si-F
C) Cl-F
D) C-F
E) C-I
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When one mole of each of the following liquids is burned, which will produce the most heat energy?
A) C 6H 14
B) C 5H 12
C) C 6H 14O
D) C 6H 12O
E) C 6H 10O 3
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following elements is the most electronegative?
A) Ne
B) Rb
C) P
D) I
E) Cl
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the strongest bond in the following group.
A) C-S
B) C-O
C) C=C
D) C≡N
E) C-F
G) All of the above
Correct Answer

verified
D
Correct Answer
verified
Multiple Choice
Acetone can be easily converted to isopropyl alcohol by addition of hydrogen to the carbon-oxygen double bond. Calculate the enthalpy of reaction using the bond energies given. 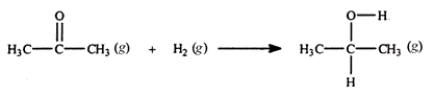
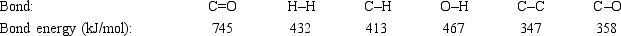
A) −484 kJ
B) −366 kJ
C) −61 kJ
D) +61 kJ
E) +366 kJ
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the compound with the lowest (i.e., least negative) lattice energy.
A) CsBr( s)
B) NaCl( s)
C) SrO( s)
D) CaO( s)
E) KBr( s)
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a covalent compound?
A) Na 2O
B) CaCl 2
C) Cl 2O
D) CsCl
E) Al 2O 3
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 61
Related Exams