A) N2O
B) NF3
C) H2Se
D) TeO3
E) CH3Cl
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molecular shape of NH2Cl as predicted by the VSEPR theory?
A) trigonal pyramidal
B) tetrahedral
C) T-shaped
D) see-saw
E) trigonal planar
G) B) and E)
Correct Answer

verified
Correct Answer
verified
True/False
The molecule AX2, where A and X are different elements, will have a dipole moment if the molecule is bent.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use VSEPR theory to predict the electron group arrangement around iodine, the central atom in the ion IF2¯.
A) octahedral
B) trigonal bipyramidal
C) tetrahedral
D) trigonal planar
E) bent
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In which one of the following species is the central atom (the first atom in the formula) an exception to the octet rule?
A) NH3
B) NH4+
C) I2
D) BH4¯
E) SF6
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict the ideal bond angles in FNO using the molecular shape given by the VSEPR theory.
A) 90
B) 109
C) 120
D) 180
E) between 120 and 180
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following molecules contains a double bond?
A) N2
B) PCl5
C) CH2O
D) C2H2
E) I2
G) B) and E)
Correct Answer

verified
Correct Answer
verified
True/False
Bond angles of 180 only occur around atoms which display linear molecular geometry.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the Lewis structure for XeO2F2 which correctly minimizes formal charges.
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the best Lewis structure for P2I4.
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) None of these structures is suitable for P2I4.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In neutral molecules, how many bonds are commonly formed by nitrogen? And how many by oxygen?
A) 5 and 6
B) 4 and 2
C) 3 and 6
D) 4 and 6
E) 3 and 2
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the correct Lewis structure for nitrogen trifluoride, NF3.
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molecular symmetry around the carbons in CCl2CH2 as predicted by the VSEPR theory?
A) linear
B) trigonal planar
C) V-shaped
D) tetrahedral
E) trigonal pyramidal
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The formal charges on Cl and O in the structure shown for the ClO¯ ion are, respectively: 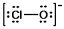
A) 0 and -1
B) -1 and 0
C) 1 and -2
D) -2 and 1
E) None of these choices is correct.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict the actual bond angle in SeCl2 using the VSEPR theory.
A) more than 120
B) between 109 and 120
C) between 90 and 109
D) exactly 90
E) less than 90
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In carbon disulfide, how many lone pairs of electrons are on each sulfur atom?
A) 0
B) 1
C) 2
D) 3
E) 4
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict the smallest actual bond angle in BrF3 using the VSEPR theory.
A) more than 120
B) exactly 120
C) between 109 and 120
D) between 90 and 109
E) less than 90
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use VSEPR theory to decide which one of the following ions and molecules is likely to be planar. (The central atom is always first in the formula.)
A) BrF3
B) H3O+
C) PCl3
D) SO42¯
E) SF4
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Essay
Using SO2 as an example, describe the sort of experimental data which might suggest that no single Lewis structure is an accurate representation of its bonding.
Correct Answer

verified
In the SO2 molecule, the two sulfur-oxyge...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
Predict the actual bond angles in SF3+ using the VSEPR theory.
A) more than 120
B) exactly 120
C) between 109 and 120
D) between 90 and 109
E) less than 90
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Showing 81 - 100 of 108
Related Exams