A) -921 kJ
B) -767 kJ
C) -460 kJ
D) -307 kJ
E) None of these choices is correct.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
The lead-acid battery is an example of a secondary battery.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about voltaic and electrolytic cells is correct?
A) The anode will definitely gain weight in a voltaic cell.
B) Oxidation occurs at the cathode of both cells.
C) The free energy change, G, is negative for the voltaic cell.
D) The electrons in the external wire flow from cathode to anode in an electrolytic cell.
E) None of these choices is correct.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the reaction: CuO(s) + H2(g) Cu(s) + H2O(l) In this reaction, which substances are the oxidant and reductant, respectively?
A) CuO and H2
B) H2 and CuO
C) CuO and Cu
D) H2O and H2
E) None of these choices is correct.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A voltaic cell prepared using zinc and iodine has the following cell notation. 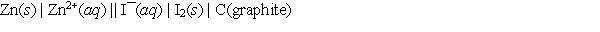 Which of the following equations correctly represents the balanced, spontaneous cell reaction?
Which of the following equations correctly represents the balanced, spontaneous cell reaction?
A) 2I¯(aq) + Zn2+(aq) I2(s) + Zn(s)
B) I2(s) + Zn(s) 2I¯(aq) + Zn2+(aq)
C) 2I¯(aq) + Zn(s) I2(s) + Zn2+(aq)
D) I2(s) + Zn2+(aq) 2I¯(aq) + Zn(s)
E) None of these choices, since graphite must be in the equation.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Essay
A concentration cell is based on the aqueous reaction  The cell consists of copper electrodes dipping into solutions of Cu2+ ions. The anions present are sulfate ions. Write the shorthand cell notation for this cell.
The cell consists of copper electrodes dipping into solutions of Cu2+ ions. The anions present are sulfate ions. Write the shorthand cell notation for this cell.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate E cell and indicate whether the overall reaction shown is spontaneous or nonspontaneous. 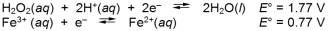 Overall reaction:
Overall reaction: 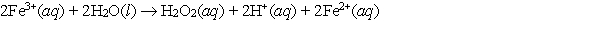
A) E cell = -1.00 V, nonspontaneous
B) E cell = -1.00 V, spontaneous
C) E cell = 1.00 V, nonspontaneous
D) E cell = 1.00 V, spontaneous
E) E cell = -0.23 V, nonspontaneous
G) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
A salt bridge provides a path for electrons to move between the anode and cathode compartments of a voltaic cell.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When metal A is placed in a solution of metal ions B2+, a reaction occurs between A and B2+, and metal ions A2+ appear in the solution. When metal B is placed in acid solution, gas bubbles form on its surface. When metal A is placed in a solution of metal ions C2+, no reaction occurs. Which of the following reactions would not occur spontaneously?
A) C(s) + 2H+(aq) H2(g) + C2+(aq)
B) C(s) + A2+(aq) A(s) + C2+(aq)
C) B(s) + C2+(aq) C(s) + B2+(aq)
D) A(s) + 2H+(aq) H2(g) + A2+(aq)
E) B(s) + 2H+(aq) H2(g) + B2+(aq)
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What product forms at the cathode during the electrolysis of molten lithium iodide?
A) Li+(l)
B) Li(l)
C) I¯(l)
D) I2(g)
E) I3¯(l)
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A voltaic cell consists of a Mn/Mn2+ electrode (E = -1.18 V) and a Fe/Fe2+ electrode (E = -0.44 V) . Calculate [Fe2+] if [Mn2+] = 0.050 M and Ecell = 0.78 V at 25 C.
A) 0.040 M
B) 0.24 M
C) 1.1 M
D) 1.8 M
E) None of these choices is correct.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A cell can be prepared from copper and tin. What is the E cell for the cell that forms from the following half-reactions? 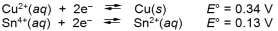
A) 0.47 V
B) 0.21 V
C) -0.21 V
D) -0.47 V
E) 0.42 V
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When the following redox equation is balanced with smallest whole number coefficients, the coefficient for zinc will be _____. 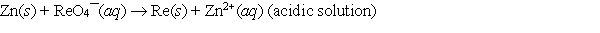
A) 2
B) 7
C) 8
D) 16
E) None of these choices is correct.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A battery is considered "dead" when:
A) Q < 1
B) Q = 1
C) Q > 1
D) Q = K
E) Q/K = 0
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When the following redox equation is balanced with smallest whole number coefficients, the coefficient for the hydrogen sulfate ion will be ______. 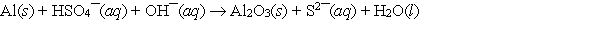
A) 1
B) 3
C) 4
D) 8
E) None of these choices is correct.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
True/False
Oxidation occurs at the cathode of a galvanic cell, but at the anode of an electrolytic cell.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
For the reaction occurring in a voltaic (galvanic) cell, G > 0.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What product forms at the anode during the electrolysis of molten NaBr?
A) Na+(l)
B) Na(l)
C) Br¯(l)
D) Br3¯(l)
E) Br2(g)
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Essay
A concentration cell is based on the aqueous reaction 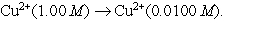 The cell consists of copper electrodes dipping into solutions of Cu2+ ions. The anions present are sulfate ions. Draw a neat diagram to represent this cell, showing and labeling all necessary components including: anode, cathode, electron flow, cation flow and anion flow.
The cell consists of copper electrodes dipping into solutions of Cu2+ ions. The anions present are sulfate ions. Draw a neat diagram to represent this cell, showing and labeling all necessary components including: anode, cathode, electron flow, cation flow and anion flow.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the reaction of iodine with manganese dioxide:
3I2(s) + 2MnO2(s) + 8OH¯(aq)  6I¯(aq) + 2MnO4¯(aq) + 4H2O(l)
The equilibrium constant for the overall reaction is 8.30 *10¯7. Calculate E cell for the reaction at 25 C.
6I¯(aq) + 2MnO4¯(aq) + 4H2O(l)
The equilibrium constant for the overall reaction is 8.30 *10¯7. Calculate E cell for the reaction at 25 C.
A) -0.36 V
B) -0.18 V
C) -0.12 V
D) -0.060 V
E) None of these choices is correct.
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Showing 81 - 100 of 102
Related Exams