A) sp2
B) sp3
C) sp3d
D) sp3d2
E) None of these choices are correct.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
True/False
Valence bond theory is able to explain why molecular oxygen is paramagnetic.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
Atoms of period 3 and beyond can undergo sp3d2 hybridization, but atoms of period 2 cannot.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Valence bond theory predicts that sulfur will use _____ hybrid orbitals in sulfur dioxide, SO2.
A) sp
B) sp2
C) sp3
D) sp3d
E) sp3d2
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Valence bond theory predicts that bromine will use _____ hybrid orbitals in BrF5.
A) sp2
B) sp3
C) sp3d
D) sp3d2
E) None of these choices are correct.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A molecule with the formula AX4 uses _________ to form its bonds.
A) sp hybrid orbitals
B) sp2 hybrid orbitals
C) sp3 hybrid orbitals
D) sp3d hybrid orbitals
E) sp3d2 hybrid orbitals
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the correct statement about 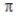 -bonds in valence bond theory.
-bonds in valence bond theory.
A) A ![]() bond is stronger than a sigma bond.
bond is stronger than a sigma bond.
B) A ![]() bond can hold 4 electrons, two above and two below the σ-bond axis.
bond can hold 4 electrons, two above and two below the σ-bond axis.
C) A ![]() carbon-carbon double bond consists of two bonds.
carbon-carbon double bond consists of two bonds.
D) A ![]() bond is the same strength as a σ bond.
bond is the same strength as a σ bond.
E) A ![]() bond between two carbon atoms restricts rotation about the C-C axis.
bond between two carbon atoms restricts rotation about the C-C axis.
G) C) and E)
Correct Answer

verified
Correct Answer
verified
True/False
Valence bond theory explains the bonding in diatomic molecules such as HCl without resorting to the use of hybrid orbitals.
B) False
Correct Answer

verified
Correct Answer
verified
Showing 41 - 48 of 48
Related Exams